  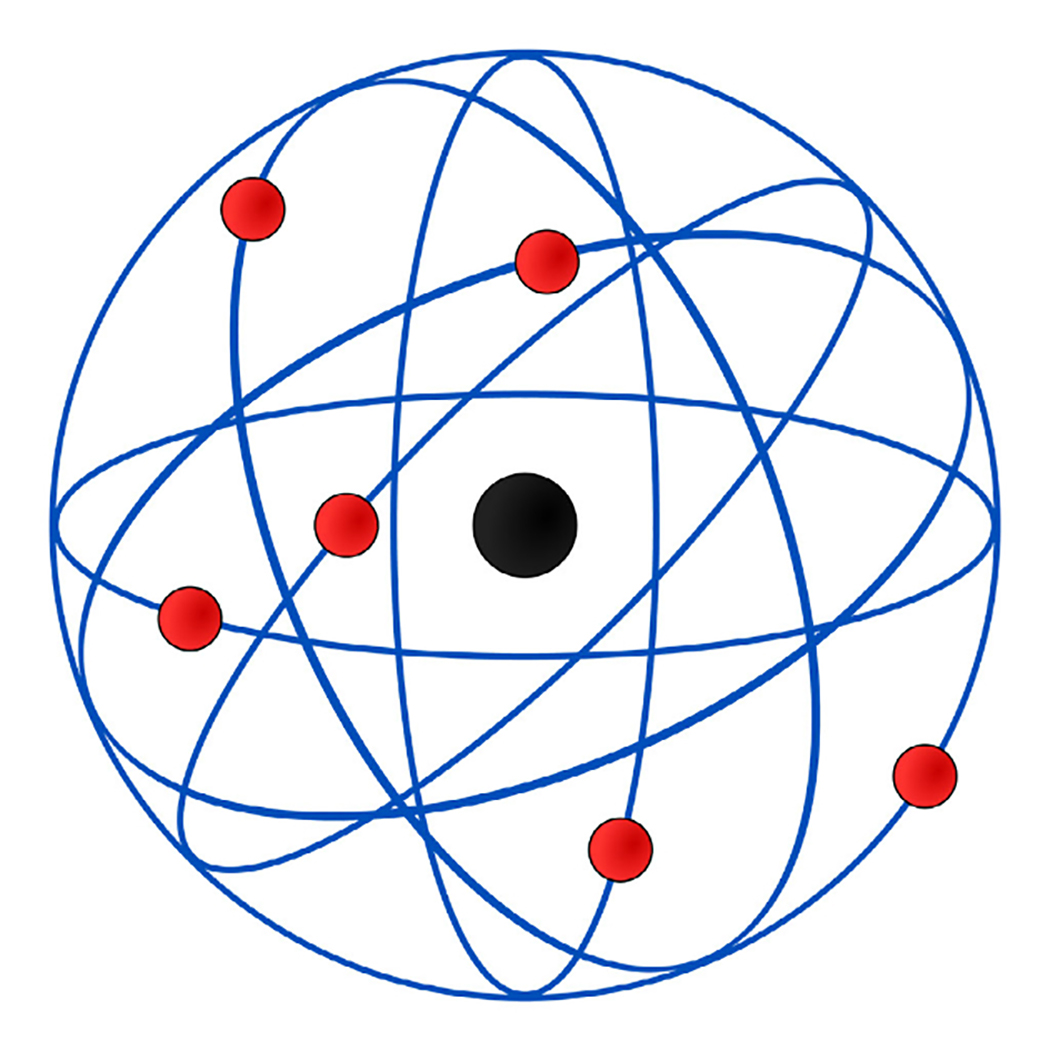
In 1900, he married Mary Georgina Newton, and they had a daughter, Eileen Mary, the next year.ĭuring his nine years in Montreal, Rutherford collaborated with the young Frederick Soddy (winner of the Nobel Prize in Chemistry in 1921) on ground-breaking research into the transmutation of elements. In 1898, Rutherford was appointed to the vacant chair of physics at McGill University in Montreal, Canada. In 1897, he was awarded a BA Research Degree and the Coutts-Trotter Studentship of Trinity College, Cambridge. During Rutherford’s investigation of radioactivity at Cambridge, he invented an ingenious detector for electromagnetic waves, and coined the terms “ alpha” and “ beta” to describe the two distinct types of radiation emitted by thorium and uranium. Thompson (soon to become the discoverer of the electron). He continued with research work at Canterbury College for a short time, receiving a BSc degree in 1894, before traveling to England in 1895 for postgraduate study at the Cavendish Laboratory at the University of Cambridge, where he studied under J. He graduated with an MA in 1893, with a double first in Mathematics and Physical Science. He was educated at Havelock School and then, at age 16, Nelson Collegiate School, before winning a scholarship to study at Canterbury College at the University of New Zealand in Wellington in 1889. He was awarded the Nobel Prize in Chemistry in 1908 “for his investigations into the disintegration of the elements, and the chemistry of radioactive substances”.Įrnest Rutherford was born on 30 August 1871 in Spring Grove (now called Brightwater) near Nelson, New Zealand, the fourth of twelve children of a Scottish farmer and an English schoolteacher. He is also credited with the discovery of the proton in 1919, and hypothesized the existence of the neutron. In 1911, he was the first to discover that atoms have a small charged nucleus surrounded by largely empty space, and are circled by tiny electrons, which became known as the Rutherford model (or planetary model) of the atom. He also had the great pleasure of seeing several of his close associates receive their own Nobel Prizes, including Rutherford in chemistry (1908) and Aston in chemistry (1922).Ernest Rutherford, 1st Baron Rutherford of Nelson was a New Zealand chemist who has become known as the “father of nuclear physics”. Thomson received various honors, including the Nobel Prize in Physics in 1906 and a knighthood in 1908. In 1913 Thomson published an influential monograph urging chemists to use the mass spectrograph in their analyses. His nonmathematical atomic theory-unlike early quantum theory-could also be used to account for chemical bonding and molecular structure (see Gilbert Newton Lewis and Irving Langmuir). Of all the physicists associated with determining the structure of the atom, Thomson remained most closely aligned to the chemical community. He was a good lecturer, encouraged his students, and devoted considerable attention to the wider problems of science teaching at university and secondary levels. Even though he was clumsy with his hands, he had a genius for designing apparatus and diagnosing its problems. In 1884 he was named to the prestigious Cavendish Professorship of Experimental Physics at Cambridge, although he had personally done very little experimental work. .jpg)
He was then recommended to Trinity College, Cambridge, where he became a mathematical physicist. Instead young Thomson attended Owens College, Manchester, which had an excellent science faculty. His father intended him to be an engineer, which in those days required an apprenticeship, but his family could not raise the necessary fee. Ironically, Thomson-great scientist and physics mentor-became a physicist by default. His assistant, Francis Aston, developed Thomson’s instrument further and with the improved version was able to discover isotopes-atoms of the same element with different atomic weights-in a large number of nonradioactive elements. Here his techniques led to the development of the mass spectrograph. Thomson’s last important experimental program focused on determining the nature of positively charged particles. His efforts to estimate the number of electrons in an atom from measurements of the scattering of light, X, beta, and gamma rays initiated the research trajectory along which his student Ernest Rutherford moved. In 1904 Thomson suggested a model of the atom as a sphere of positive matter in which electrons are positioned by electrostatic forces. Structure of the Atom and Mass Spectrography 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
